Our research focuses on supramolecular chemistry, particularly self-assembly.
Specifically, we are interested in using relatively weak non-covalent interactions such as hydrogen bonding and halogen bonding to assemble complex/interesting/potentially useful 3D architectures. This encompasses aspects of host–guest chemistry, self–assembly and crystal engineering. Most of our work is focused on two major areas: supramolecular frameworks and hydrazone cages. Specific areas of research are described in more detail below, with some selected references provided.
Supramolecular frameworks:
We have developed a (relatively) general route to a large family of supramolecular frameworks assembled through charge-assisted amidinium···carboxylate hydrogen bonds. These are typically prepared in water, and are surprisingly robust (including to extended heating in polar organic solvents and water). Working with Christian Doonan and colleagues at the University of Adelaide, we have demonstrated that these frameworks can be used to encapsulate and stabilise enzymes. Working with Jonathan Foster at the University of Sheffield, we have shown that 2D hydrogen bonded systems can be exfoliated into single layer nanosheets. We have also recently demonstrated an ion-pairing route to porous halogen bonded frameworks.
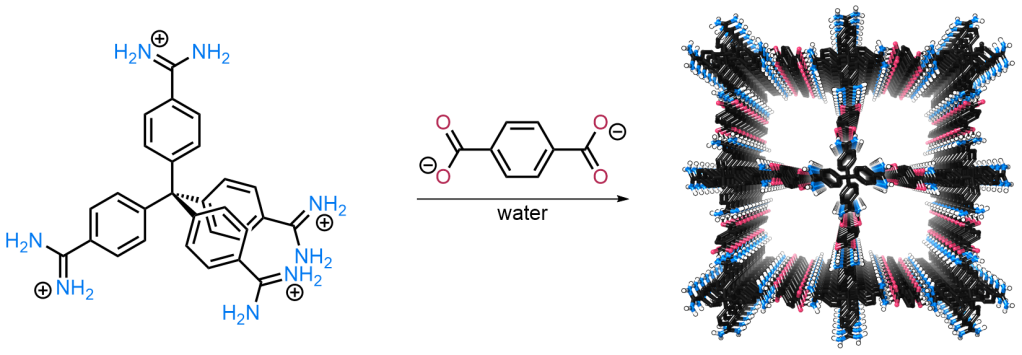
Initial framework: Chem. Sci. 2017, 3019
Enzyme encapsulation: J. Am. Chem. Soc. 2019, 14298
Hydrogen bonded nanosheets: Chem. Sci. 2021, 3322
Mechanisms of water sorption and structural rearrangements: Chem. Eur. J. 2022, e202201929, Angew. Chem. Int. Ed. 2023, e202212962
Selective anion precipitants based on amidinium···anion interactions: Angew. Chem. Int. Ed. 2023, e2022218360
Porous halogen bonded frameworks through ion pairing: Angew. Chem. Int. Ed. 2025, e202422197
Review of supramolecular frameworks trying to sort out nomenclature and characterisation standards: Chem. Soc. Rev. 2025, accepted
Cages:
We have recently developed a family of robust organic cages that can be prepared on large scales (14 g) in high yields through short syntheses. We are working with Dr Annie Colebatch’s group (ANU) to investigate metalating these cages and exploring their use for guest binding, sensing and catalysis. Our initial studies have shown that metalation can lead to low symmetry metallocages.
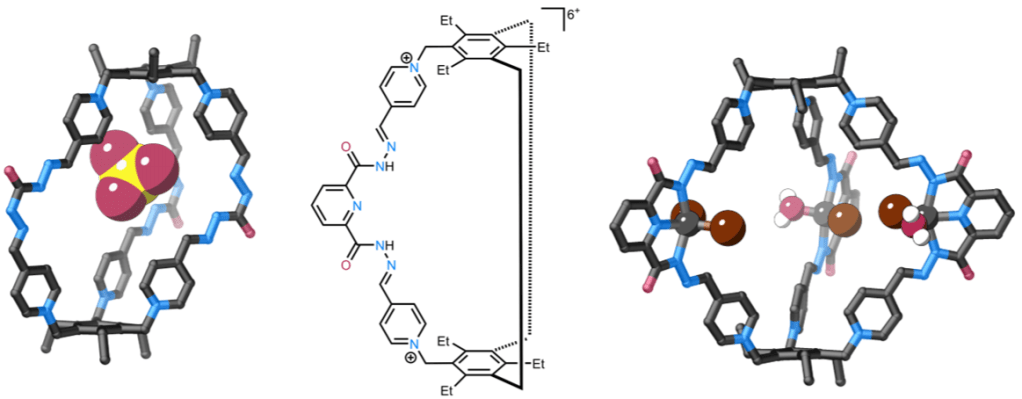
Large scale synthesis of cages (up to 14 g) for selective sulfate recognition in water: J. Am. Chem. Soc. 2024, 27127
Low symmetry cages upon metalation: Angew. Chem. Int. Ed. 2025, e202513159
Other things:
Over the last few years, we have also worked in a few different areas that are a bit different from our current main focuses. Some representative papers are listed below.
Anti-electrostatic hydrogen bonds: CrystEngComm, 2019, 4855, Chem. Soc. Rev. 2020, 7893
O–H···anion interactions: Org. Biomol. Chem. 2021, 2794, Chem. Asian J. 2024, e202401121
Rotaxanes for anion recognition: Chem. Eur. J. 2022, e202200389
Pillar[6]arene synthesis: J. Org. Chem. 2023, 8310, J. Org. Chem. 2024, 1397
Alkyne cages, including for post-synthetic metalation: Chem. Eur. J. 2022, e202200958, Supramol. Chem. 2024, 326